Sensors and Cameras for Plant Phenotyping and Seed Testing
LemnaTec provides a wide range of approved sensors for all aspects of plant phenotyping and seed testing. Sensors and accessories, e.g. lamps and background materials, are tested in real research scenarios and the results are documented and supported by case studies and references.
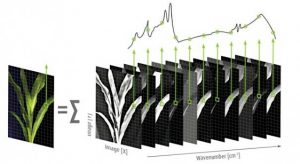
The application of sensors for phenotyping aims at capturing dimensions, morphologies and colours of the samples. To address physiology, non-visible radiation such as near-infrared or fluorescence, is captured in addition. Spectral imaging and 3D scans deliver detailed insights in physiological and structural properties.
Data from sensors can be translated into traits or biological properties using appropriate mathematical models. Such models establish links between non-invasive measurements and actual plant characteristics.
| Sensor | Recorded parameters | Information to derive from parameters |
|---|---|---|
| Visible light camera | Reflectance in visible light spectrum (colour image) | Counts, dimensions, texture, colour; growth and developmental features, stress responses |
| Fluorescence camera (with corresponding excitation light and filter) | Intensity and distribution of fluorescence light | Presence and distributions of fluorescencent pigments; stress and senescence |
| Camera for chlorophyll fluorescence dynamics (Kautsky/PAM) | Photosystem II-related parameters | Photosynthetic capacity and activity; stress, pathogen responses |
| NIR camera with 1450 nm filter | Water-content related NIR signal | Tissue moisture; water stress responses |
| IR camera | Surface heat emission | Plant temperatures, transpiration |
| Hyper-/multispectral camera | Spectrally resolved reflectance | Physiological parameters, vegetation indices |
| Laser scanner | Point clouds | 3D surface, height map, inclination map, convex hull; growth and developmental features |
| High-sensitivity camera | Bioluminescence signals | Physiological features e.g., gene expression or plant-microbe interaction |